What is document control process? It is the foundation of any efficient and compliant business, ensuring documents are created, reviewed, approved, and maintained accurately throughout their lifecycle. In this guide, DIGI-TEXX will help you understand how a document control process works, its key benefits, requirements, and essential steps to build a streamlined system that improves accuracy, reduces errors, and supports compliance across your organization.

>>> See more:
- Data Validation vs Verification: Key Differences, Examples & Benefits
- Document Management Outsourcing: Benefits, Services & Companies
- Medical Claims Processing Outsourcing For Healthcare Providers
What Is The Document Control Process?
The document control process is the formal path that documents travel through your company based on clearly defined rules and regulations. It maintains each document one, approved master copy and a reproducible history to ensure consistency, accuracy, and accountability.
That is, a document control procedure defines the precise steps involved in handling documents from preparation to disposal. This includes identification of types of documents utilized, proper identification assignment, tracking of changes to versions, and control of controlled and uncontrolled copies.
The procedure also includes document review, approval routines, secure storage, access, and long-term retention policies. It also includes change control procedures, document security practices, and submission and approval guidelines. All these phases collect to provide a sound, compliant, and efficient document handling system.
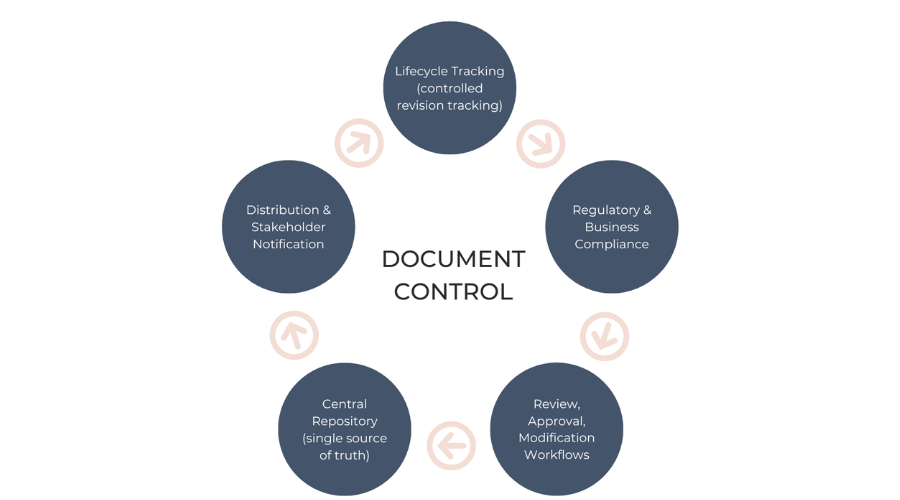
The document control process is the formal path that documents travel through your company
Benefits Of An Efficient Document Control Process
Having an efficient document control system is advantageous to both small and big businesses.
- Simplified and quicker documents: An efficient document control system ensures that employees can quickly find and obtain the documents they need, thereby saving time and enhancing responsiveness within teams.
- Less error and more accuracy: Automating the document control process enables organizations to bypass human mistakes resulting from handling outdated or unapproved documents, which are most commonly responsible for compliance failure.
- Less operational costs: Automation of a portion of the document control system saves organizations labor dollars, printing dollars, and storage dollars—ultimately saving them money.
- More operations and saved time: Organized workflows save time spent searching through files or redoing things due to outdated content. An effective document control process guarantees that operations go efficiently.
- Greater protection of data and access: The system guarantees that only approved individuals can open or modify confidential files, keeping company secrets safe and less likely to leak or be unauthorizedly edited.
- Improved compliance and audit readiness: An effective document control system enables organizations to achieve compliance and clear audits by version control, valid approvals, and traceability.
- Improved quality assurance and client satisfaction: With compliant documents, teams produce the right work consistently, which improves quality and inspires clients’ trust. Document control process ensures that there is a base of reliability and professionalism in place.
>>> See more:
- What is Automated Document Processing and How Does It Work?
- Administrative Support Outsourcing: Services, Top Providers & How to Choose
- Data Labeling Service: What It Is, Types & Benefits For AI

What Are The Requirements For Document Control In A Document Management System?
Document control requirements in a document management system are defined by international standards, regulations, and industry guidelines. These frameworks ensure that documents are accurate, secure, and properly managed throughout their lifecycle, especially in highly regulated sectors like life sciences.
Key standards and regulations include:
- ISO 9001:2015: Requires organizations to control documented information to ensure availability, accuracy, and protection. It also defines how documents are created, accessed, updated, stored, and disposed of.
- ISO 13485:2016: Focuses on strict document and record control, including approval, version tracking, and ensuring the correct documents are available at the point of use.
- ISO 15189:2022: Requires laboratories to manage both internal and external documents with clear identification, regular review, and controlled access, along with proper record retention practices.
- 21 CFR Part 820: Mandates formal procedures for document review, approval, distribution, and change control within medical device quality systems.
- EudraLex Volume 4 GMP Part I: Emphasizes clear documentation structure, controlled creation and updates, and maintaining document integrity throughout the retention period.
- ICH Q7: Requires full control over document approval, revision history, and lifecycle management to ensure traceability and compliance.
Key Components Of A Document Control Process
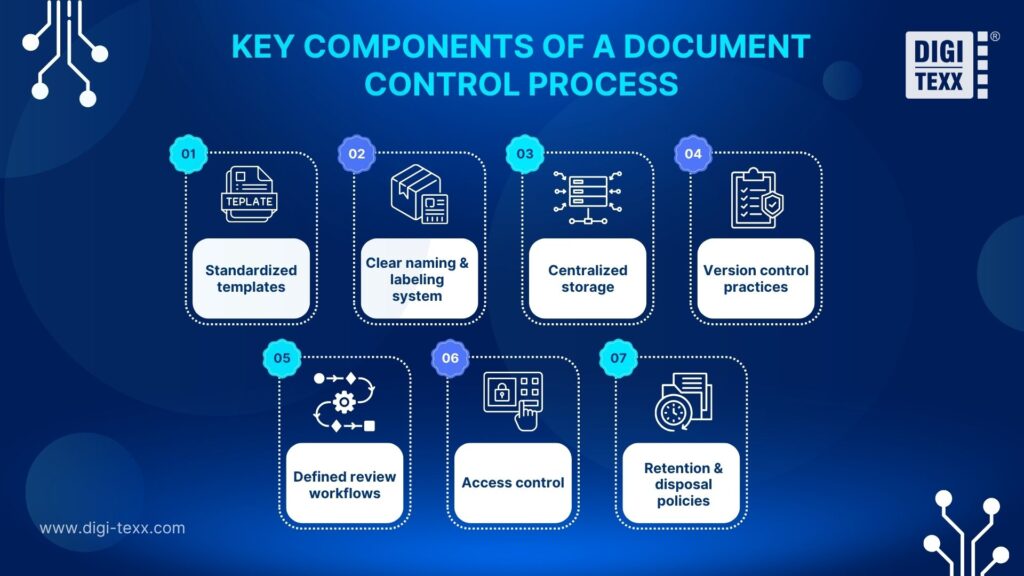
Document control combines clear processes, the right technology, and proper governance to keep documents accurate, up to date, and accessible only to authorized users. To make it work effectively, organizations need defined guidelines for every stage of the document lifecycle, from creation to final disposal.
These guidelines help standardize how documents are created, stored, edited, and retired. While each organization may design its system differently, a strong document control program typically includes:
- Standardized templates: Ensure consistency in format and content across all documents.
- Clear naming and labeling system: Make documents easy to identify, search, and manage.
- Centralized storage: Keep all documents in one secure repository for better access and version control.
- Version control practices: Track changes and ensure everyone works on the latest version.
- Defined review workflows: Streamline how documents are reviewed and approved across teams.
- Access control: Restrict or grant access based on roles to protect sensitive information.
- Retention and disposal policies: Set rules for how long documents are kept and how they are securely removed.
With the right tools and well-defined processes, organizations can manage documents efficiently at every stage. It is equally important to train teams on these practices to ensure consistent adoption across the business.
>>> See more:
- What Are the 6 Steps of Data Analysis? A Practical Guide For Businesses
- Professional E-commerce Data Entry Services by DIGI-TEXX
- Invoice Processing Outsourcing Services For US Businesses
What Types Of Documents Need To Be Controlled?
Document control applies to a wide range of files, depending on industry standards and internal policies. In most quality management systems, it ensures that critical documents remain accurate, consistent, and compliant throughout their lifecycle.
Common document types that require control include:
- Quality manuals: Define the overall quality system and compliance framework.
- Policies and objectives: Outline commitments and measurable quality goals.
- Standard operating procedures (SOPs): Guide how tasks and processes are performed.
- Specifications: Detail technical or product requirements for quality evaluation.
- Forms and templates: Standardize data collection and document creation.
- Records: Provide proof that processes and actions have been completed correctly.
Together, these documents form the backbone of operational consistency, helping organizations maintain quality, traceability, and regulatory compliance.
What Is The Difference Between Document Control & Document Management?
Document management and document control are closely related but serve different purposes within an organization.
Document management is the broader discipline that handles documents throughout their lifecycle. It focuses on storing, organizing, retrieving, and sharing documents efficiently. A document management system (DMS) enables users to create, access, and collaborate on documents while ensuring they are easy to find and securely stored.
Document control, by contrast, is a subset of document management that focuses on critical documents affecting operations, quality, and compliance. It involves structured processes such as review, approval, version control, change tracking, distribution, and archiving. The goal is to ensure that only the most current and approved versions are in use, while outdated versions are properly managed.
The key difference lies in scope. Document management covers all documents for operational efficiency, while document control applies strict governance to selected documents that require accuracy, traceability, and regulatory compliance.
In practice, both functions are interconnected. Every controlled document is managed, but not all managed documents require control. Document control relies on document management as its foundation to maintain consistency and compliance across the organization.

>>> See more:
- Invoice Reconciliation Process: Step-By-Step Guide & Key Steps
- Professional E-commerce Data Entry Services by DIGI-TEXX
- Back Office Service: Definition, Examples, Benefits & Outsourcing 2026
7 Essential Steps To Implement A Document Control System
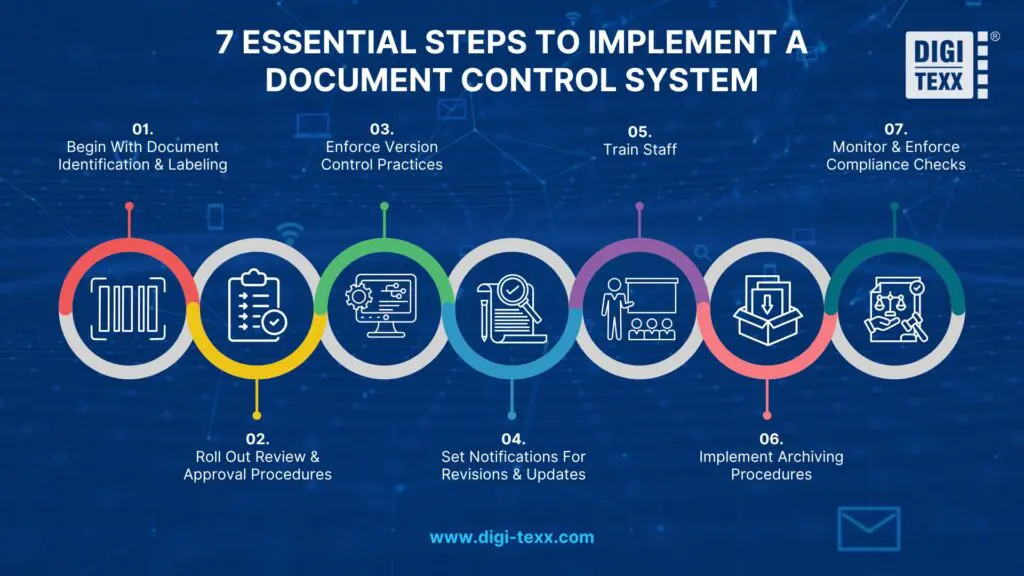
This stage is implementation-focused, outlining how to build and execute an effective document control process in practice. Below are seven essential steps to implement a scalable and compliant system:
1. Begin With Document Identification & Labeling
Start by defining clear naming conventions and document categories. Assign unique identifiers to support accurate tracking throughout the document lifecycle.
- Use tags or metadata for easier filtering and retrieval.
- Standardize naming across the organization.
- Communicate guidelines clearly to avoid inconsistencies.
2. Roll Out Review & Approval Procedures
Establish a structured workflow with clearly defined roles and responsibilities.
- The author drafts the document.
- Reviewers revise and provide feedback.
- Authorized personnel give final approval.
- Set deadlines, reminders, and maintain audit trails.
3. Enforce Version Control Practices
Version control is a critical part of any document control process.
- Use naming conventions to distinguish versions (v1.0, v1.1).
- Indicate latest vs outdated documents clearly.
- Add notes to compare changes between versions.
- Ensure every version is reviewed and approved.
- Archive all outdated versions properly.
4. Set Notifications For Revisions & Updates
Automate notifications to keep stakeholders informed of any document changes.
- Enable alerts for updates and revisions.
- Ensure timely review and action.
- Support audit readiness and compliance tracking.
5. Train Staff
Ensure all employees understand their roles in the document control process.
- Provide training on labeling, approvals, and version tracking.
- Offer hands-on guidance for DMS tools.
- Maintain ongoing support and collect feedback.
6. Implement Archiving Procedures
Define how long documents should be retained and when they become inactive.
- Set retention periods based on regulations.
- Store archived documents securely.
- Apply strict access controls to sensitive data.
7. Monitor & Enforce Compliance Checks
Continuously evaluate the system to ensure effectiveness and compliance.
- Conduct regular audits and performance reviews.
- Identify gaps and areas for improvement.
- Report findings and enforce corrective actions.
>>> See more:
- Back Office Support Services To Streamline Operations & Boost Efficiency
- Top 10 Data Entry Outsourcing Companies to Hire in 2026
- Document Indexing Services For Fast Search, Accurate Data & Easy Retrieval
Which Industries Benefit Most From A Document Control System?
Industries that manage complex documentation and strict compliance benefit most from a document control process.
Manufacturing
Manufacturers rely on detailed documentation for product specifications, quality control, safety procedures, and supply chain operations. A document control system keeps everything consistent, traceable, and aligned with industry standards.
Healthcare
Hospitals, clinics, and healthcare providers handle large volumes of patient records, medical reports, and compliance documents every day. A document control system helps ensure data accuracy, protect patient confidentiality, and maintain strict compliance with healthcare regulations.
Legal & Professional Services
Law firms and service providers handle contracts, case files, and client records. A document control system helps organize information, reduce errors, and improve document accessibility.
Finance & Banking
Financial organizations deal with sensitive data, contracts, and regulatory documents. A document control system strengthens data security, simplifies audits, and supports compliance efforts.
Engineering & Construction
From blueprints to technical drawings and safety guidelines, project teams need access to accurate and up to date documents. A document control system improves collaboration and ensures everyone works from the latest version.
Government & Public Sector
Public organizations manage a wide range of administrative, legal, and regulatory documents. A document control system supports transparency, accountability, and compliance with legal standards.
Energy & Utilities
With complex regulations and technical requirements, the energy sector depends on accurate documentation for safety, engineering, and environmental compliance. A document control system improves efficiency and regulatory alignment.
Pharmaceuticals
Pharmaceutical companies must manage complex documentation across drug development, clinical trials, and production. A document control system helps organize, track, and maintain compliance with strict regulatory requirements.
Aerospace & Defense
This industry demands precise documentation for design, maintenance, and safety compliance. A document control system ensures accuracy, consistency, and adherence to rigorous safety standards.
Information Technology
IT teams manage project documentation, development workflows, coding standards, and system changes. A document control system helps keep everything structured, updated, and easy to access.
>>> See more:
- Ecommerce Back Office Support Services For Fast Growth
- Boost Efficiency With Insurance Back Office Support Services
- Intelligent Document Processing Services: Benefits, Use Cases & How It Works
When Should Your Business Implement A Document Control Process?
A document control process becomes essential when your business needs to keep information accurate, reduce errors, meet compliance requirements, and maintain smooth, consistent workflows. This is especially important in highly regulated industries such as healthcare, finance, manufacturing, and engineering, where poor document handling can lead to serious risks.
More broadly, if your team is struggling with manual processes, scattered files, or frequent mistakes, it’s a clear sign that it’s time to implement a document control process. By standardizing and automating how documents are managed, businesses can improve efficiency, reduce risk, and ensure better control over critical information.
FAQs About Document Control Process
Why Is A Document Control Process Important?
A document control process ensures documents are accurate, up to date, and accessible to the right people. It helps prevent errors caused by outdated information, improves workflow efficiency, and supports compliance with regulations through proper version control and audit trails. It also enhances data security by restricting access to authorized users and ensures full traceability of changes.
How Does A Document Control Process Work?
A document control process works by managing documents through a structured lifecycle to ensure accuracy, consistency, and compliance. It typically includes creating and reviewing documents, assigning versions, approving for use, distributing to the right users, controlling access, tracking changes, and regularly updating or archiving outdated versions.
If you want to optimize your organization’s document handling through automation, security, and compliance as the cornerstones, DIGI-TEXX is the reliable partner who can assist you in getting it right. With extensive hands-on experience in digital document transformation and smart document processing, DIGI-TEXX provides scalable solutions specific to the requirements of your industry—enabling you to optimize your process without compromising complete control and traceability of your documents.
DIGI-TEXX Contact Information:
🌐 Website: https://digi-texx.com/
📞 Hotline: +84 28 3715 5325
✉️ Email: [email protected]
🏢 Address:
- Headquarters: Anna Building, QTSC, Trung My Tay Ward
- Office 1: German House, 33 Le Duan, Saigon Ward
- Office 2: DIGI-TEXX Building, 477-479 An Duong Vuong, Binh Phu Ward
- Office 3: Innovation Solution Center, ISC Hau Giang, 198 19 Thang 8 street, Vi Tan Ward
Reference:
- International Organization for Standardization. ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes. https://www.iso.org/standard/59752.html
- National Archives and Records Administration. Records Management. https://www.archives.gov/records-mgmt


